Sep 20 2017
Fluorescence microscopy offers Researchers unbelievable power to illuminate the smallest structures and capture the real-time activities of live cells by tagging biological molecules with a veritable rainbow of fluorescent dyes. However, this power comes at a cost: The technology can be time-consuming and expensive, and has so far resisted attempts at automation.
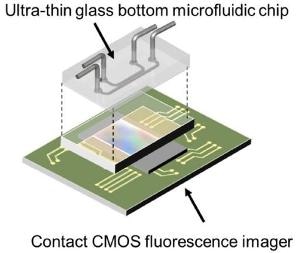 A diagram of the on-chip fluorescence imaging platform showing how the ultra-thin glass bottom microfluidic chip sits on top of the contact CMOS fluorescence imager. (Credit: Takehara et al.)
A diagram of the on-chip fluorescence imaging platform showing how the ultra-thin glass bottom microfluidic chip sits on top of the contact CMOS fluorescence imager. (Credit: Takehara et al.)
However, this situation may be changing with the introduction of microfluid chip-based platforms. A team of Japanese Researchers have developed one such recently developed platform. Their system allows Scientists to quickly image fluorescent cells grown within the chip using a CMOS image sensor, the same technology available in the camera of a smartphone. The latest system, described this week in AIP Advances, from AIP Publishing, has several promising uses in biomedical research.
“Conventional tabletop-type optical microscopes are powerful tools for researchers, but they are not truly adequate for fully automated systems because of the expense and the necessity of well-trained technicians,” said Hiroaki Takehara, who studies automated cell processing devices at the University of Tokyo and is one of the study’s Authors.
Takehara teamed up with Co-author Jun Ohta of the Nara Institute of Science and Technology, an expert in CMOS image sensor technology, in order to develop an on-chip system.
Previously, other groups have developed chip-based fluorescent microscopy systems, but those setups required the sample to sit on the image sensor chip directly, which introduces the risk of cross-contamination. These systems cannot be actually high-throughput as the sensor chips must be washed in between use.
Takehara and colleagues developed disposable chips to solve these challenges. The chip consists of microfluidic channels exclusively designed for culturing cells and the introduction of drugs, culture media and other biological molecules. The chip has an ultra-thin glass bottom that reduces the distance between the cells and the contact sensor below. A CMOS image sensor identifies the fluorescence emitted by the cells, converts it into an electronic signal and then reconstructs the image.
The Researchers grew cells containing fluorescent dyes in their nuclei within the microchannels in order to demonstrate the effectiveness of their system. When they exposed cells to EGF (endothelial growth factor), which causes cell proliferation, the cultures gave off a more strong fluorescence signal than cultures that were not treated with EGF, signifying that the sensor discovered cell growth.
The Authors accept that the on-chip fluorescence microscopy platform produces images with poorer spatial resolution than those of traditional fluorescence microscopes, but provides the advantage of being compatible with completely automated systems. The affordability and small size of the platform also make it attractive for use in implantable devices for not only measuring glucose but even brain activity.
In future, Takehara is planning to investigate the use of the platform for monitoring stem cell production for screening new drugs and for use in regenerative medicine.
The excessive cost of developing novel pharmaceutical drugs and the urgent requirement for [affordable] screening technology has become a pressing issue. A fully automated system, from sample handling to detection, without the necessity of well-trained technicians is a key technology, and serves a pivotal role in the development of cell-based cost-effective screening.
Hiroaki Takehara, Student, the University of Tokyo and is one of the study’s Authors