As one of the most widely used materials in society, it is thus no surprise that global rates of steel production continue to rise. Production rates have reached ~1,550 Mt/y as a result of this growing demand.
Blast furnaces are used to make the majority of raw iron, which necessitates the implementation of high-quality coke and high temperatures. An alternative process, namely the direct reduction of iron (DRI), has lower energy demands and fuel quality. DRI makes up ~75 Mt/y of iron.
DRI results in the production of spongy iron, which is processed further by sintering. This process obtains more easily transportable and compact hot briquetted iron (HBI), which is then molten by arc furnaces, for example, to create steel alloys.
Controlling the Quality of Direct Reduced Iron and Hot Briquetted Iron
It is necessary to measure the metallization (total iron Fe(total), metallic iron Fe(metallic)) and the Fe2+ content in order to ensure the quality of the starting iron ore sinters and the resulting DRI and HBI. These methods are typically not accessible by common spectroscopic methods. Time-intensive wet chemistry is thus utilized instead.

Figure 1. ARL EQUINOX 100 X-ray Diffractometer. Fast, real-time, and convenient. Image Credit: Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers
However, a new route to obtain these measurements has been obtained by Rietveld refinements of easily obtainable X-Ray diffraction (XRD) patterns. Within a typical process control environment, there is also the possibility to conveniently determine both FeO(total) content and metallization.

Image Credit: Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers
About the ARL EQUINOX 100 X-Ray Diffractometer
A custom-designed Cu (50 W) or Co (15 W) micro-focus tube with mirror optics is utilized in the Thermo Scientific™ ARL™ EQUINOX 100 X-Ray Diffractometer. External water chillers or other peripheral infrastructure is no longer needed. The low-wattage system allows the instrument to be easily transported from the laboratory to the field or between labs.
Very fast data collection times are facilitated by the ARL EQUINOX 100 Diffractometer (cf. Figure 1) compared to other conventional diffractometers. This happens due to the unique curved position sensitive detector (CPS), which can concurrently measure all diffraction peaks in real-time.
Case Study
Using Co Kα radiation for five minutes, HBI and fine-powdered sinter samples were measured in reflection geometry. With the MDI JADE 2010 and the ICDD PDF4+ database, qualitative and quantitative analysis (Rietveld refinement) was carried out.
Results
The amount of the important phases that facilitate the calculation of FeO(total) concentration and metallization in the DRI process (cf. Table 1) are yielded by the Rietveld refinements of sinter (cf. Figure 2) and HBI (cf. Figure 3).
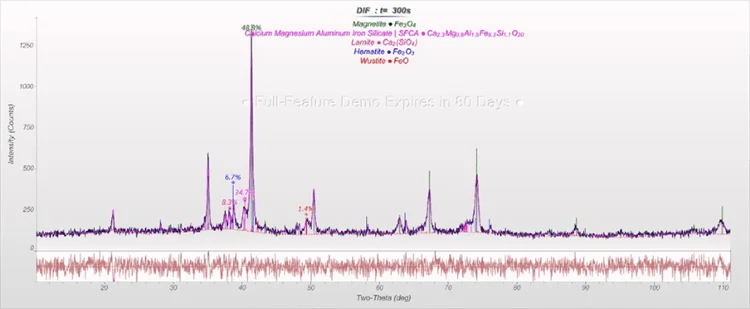
Figure 2. Rietveld refinement of a sinter sample. Image Credit: Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers
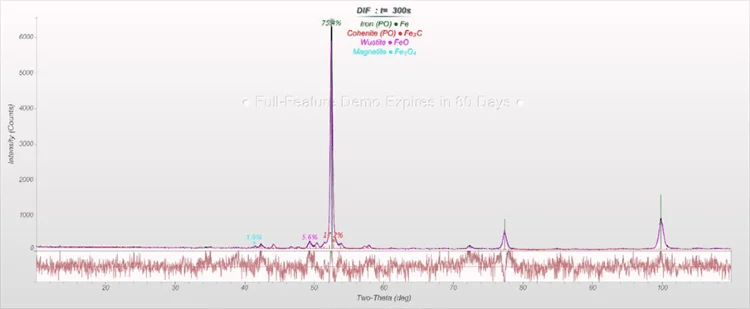
Figure 3. Rietveld refinement of an HBI sample. Image Credit: Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers
FeO(total) = 16.5 percent, and the HBI sample shows Fe(total) = 97.1 percent, Fe(metallic) = 91.4 percent (with Fe3C part, frequently included in the calculation as metallic Fe) and C = 1.2 percent are all contained within the sinter sample.
The values are in the range that is expected with samples such as those used in this article.
Table 1. Results of the Rietveld refinements of sinter and HBI samples. Source: Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers
| Phase (in wt%) |
Sinter |
HBI |
| Fe |
- |
75.4 |
| FeO |
1.4 |
5.6 |
| Fe3O4 |
48.8 |
1.9 |
| Fe2O3 |
6.7 |
0 |
| Fe3C |
- |
17.2 |
| SFCA* |
34.7 |
- |
| Larnite (Ca2SiO4) |
8.3 |
- |
*silico-ferrite of calcium and aluminum
Summary
An appropriate solution to expediently determine FeO(total), Fe(total) and Fe(metallic) in sinter and HBI samples is the use of the ARL EQUINOX 100 Benchtop X-Ray Diffractometer alongside the MDI JADE 2010 software suite and ICDD pdf4+ database.
It is essential that an accessible method using a rugged instrument, such as the ARL EQUINOX 100 Benchtop XRD, is available, given that these types of analyses are frequently done in production lines for routine operation by the users.
In order to improve productivity and quality, the data processing process can be entirely automated within this infrastructure, which will reduce the chance of manual errors occurring and optimize the workflow.
Acknowledgments
This article was produced from materials originally written by Dr. Simon Welzmiller from Thermo Fisher Scientific.

This information has been sourced, reviewed and adapted from materials provided by Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers.
For more information on this source, please visit Thermo Fisher Scientific - Elemental Analyzers and Phase Analyzers.