Image Credit: Sergei Drozd/Shutterstock.com
Gaining a deeper level of understanding of a protein’s structure opens the door to fine-grain detail on how the protein functions and what its purpose may be. It also gives scientists the opportunity to develop hypotheses on how they affect the protein, as well as how to control and modify it. This means that in knowing a protein’s structure, scientists can work on creating site-directed mutations with the purpose of changing the protein’s function.
This capability is vital to the development of new therapeutic approaches for a wide range of diseases due to the fact that specific proteins are often implicated in the development and progression of both physical and psychological illnesses.
Many methods have been established to determine protein structure, such as X-ray crystallography and NMR spectroscopy. Here, we will discuss how electron microscopy is emerging as a dominant method of establishing protein structure.
Protein structure
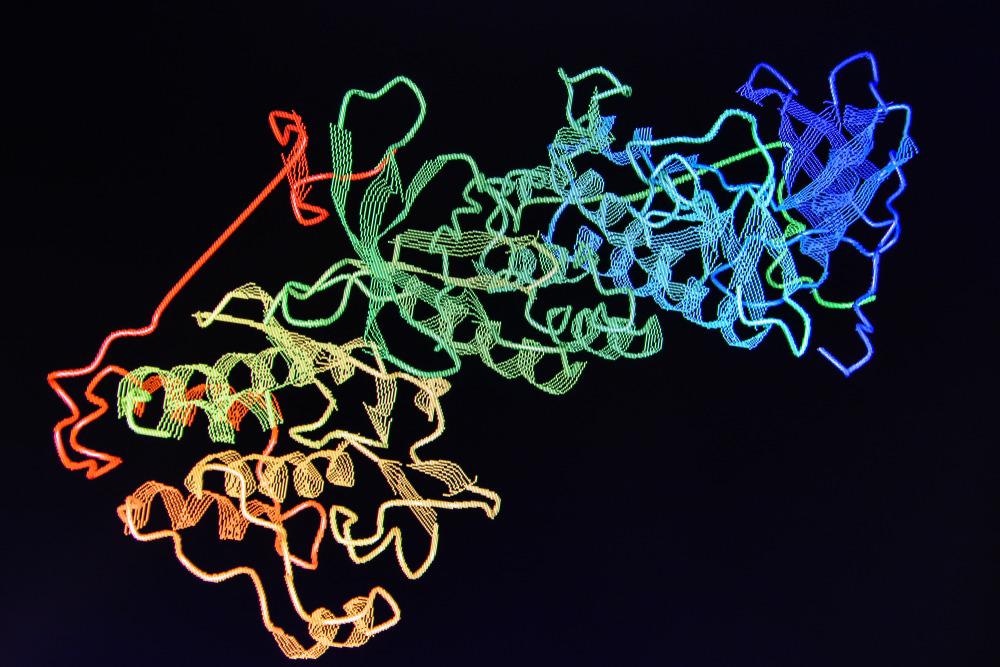
The structure of proteins is complex. Any given protein has four levels of structure: primary, secondary, tertiary, and quaternary. A protein’s primary structure is related to its amino acid sequence which causes the folding and intramolecular bonding along the amino acid chain which produces the protein’s three-dimensional shape, unique to the protein.
The secondary structure is created by the alpha helices and beta sheets that are produced from the hydrogen bonding of the protein’s amino groups and carboxyl groups, and the tertiary structure is produced from the pattern of formations and folds within a single linear chain of amino acids. Finally, the protein’s quaternary structure is created by the macromolecules with multiple polypeptide chains or subunits.
The activity of proteins is essential to human life, and its structure dictates much about how an individual protein behaves. Given that protein structure is complex, methods that allow us to accurately determine the nature of protein structure are vital to developing our understanding of how specific proteins are linked with human function as well as disease and illness.
Electron microscopy as a method of determining protein structure
X-ray crystallography is the most popular technique used for determining the structure of proteins. However, it has its limitations, and now, a revolutionary technique known as cryo-electron microscopy is seeing rapid widespread adoption and looks set to overtake X-ray crystallography in the investigation of protein and biological molecule structure.
For many decades, X-ray crystallography has been used by structural biologists to study protein structure. The technique crystallizes proteins and then exposes them to X-rays which are used to reconstruct the protein’s shape using patterns of diffracted light. However, the process can take months or years to complete due to the time taken to crystallize the structures.
Cryo-electron microscopy solves this problem in that it does not require crystallization, although its use in the field was hindered by the initially low-resolution images it produced. In comparison to X-ray crystallography, cryo-electron microscopy flash freezes proteins rapidly, without the need to wait for the lengthy crystallization process. Electrons are then introduced to the frozen samples, where they are used to reconstruct the structure of the protein.
The use of cryo-electron microscopy in determining protein structures is growing at an exponential rate. In 2020, a database that compiles structures of protein and molecules determined by cryo-electron microscopy surpassed its 10,000th entry.
The growing popularity of cryo-electron microscopy in recent years is mostly due to the rapidly rising number of cryo-electron microscopes in labs across the globe. Particularly, following significant advancements in hardware and software in 2012 and 20123 that enhanced the sensitivity of cryo-electron microscopes, enabling them to produce more detailed imagery.
Initially, only a small number of labs around the world had access to cryo-electron microscopes. However, as more and more detailed maps of protein structures were produced with the equipment, the technique rapidly began to gain popularity. By 2024, it is estimated that cryo-electron microscopy will have overtaken X-ray crystallography in terms of its use in determining protein structure.
The future of cryo-electron microscopy in determining protein structure
Cryo-electron microscopy has already determined the protein structures of numerous simple proteins. Now, scientists are beginning to use the technique to investigate more complex problems. It is predicted that the method will be key to investigating membrane-bound proteins that are vital to the development of new drug targets.
One factor that could potentially hinder the growth of cryo-electron microscopy in the future is its cost. Equipment is still relatively expensive to buy and run and experts believe these costs must come down before cryo-electron microscopy’s potential can be fully realized.
References and Further Reading
Callaway, E., 2020. Revolutionary cryo-EM is taking over structural biology. Nature, 578(7794), pp.201-201. https://www.nature.com/articles/d41586-020-00341-9
Jonic, S. and Vénien-Bryan, C., 2009. Protein structure determination by electron cryo-microscopy. Current Opinion in Pharmacology, 9(5), pp.636-642. https://www.sciencedirect.com/science/article/abs/pii/S1471489209000484
Muñoz, I., Blanco, F. and Montoya, G., 2008. On the relevance of defining protein structures in cancer research. Clinical and Translational Oncology, 10(4), pp.204-212. https://pubmed.ncbi.nlm.nih.gov/18411193/
Protein Structure. Nature Education. Available at: https://www.nature.com/scitable/topicpage/protein-structure-14122136/
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.