Dec 4 2014
While aptamers—single-stranded nucleic acids—offer promise as drug-delivery tools for cancer treatment, it has proven difficult to get them to tumor cells at therapeutic levels.
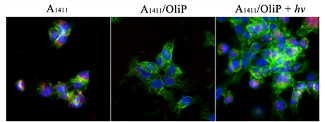 Short snippets of DNA called aptamers (red) readily get into cancer cells (green and blue) on their own (left panel). They can't penetrate cells when stuck to an oligonucleotide (center), but regain the ability when the oligonucleotide's bonds are broken by light (right). Credit:Images courtesy of Lele Li, Ph.D.
Short snippets of DNA called aptamers (red) readily get into cancer cells (green and blue) on their own (left panel). They can't penetrate cells when stuck to an oligonucleotide (center), but regain the ability when the oligonucleotide's bonds are broken by light (right). Credit:Images courtesy of Lele Li, Ph.D.
Now, researchers in the laboratories of Robert Langer, the David H. Koch Institute Professor and member of the Koch Institute, and Daniel S. Kohane, director of the Laboratory for Biomaterials and Drug Delivery at Boston Children’s Hospital, describe in the Proceedings of the National Academy of Sciences how a new light-triggered strategy can provide more accurate control over where aptamers accumulate.
Aptamers have various applications in cancer imaging, diagnostics, and therapeutics. They can be easily synthesized in the lab, and they can demonstrate high affinity and selectivity toward targets such as molecules, proteins, and cells.
While tumor-targeting aptamers would ideally accumulate in tumor tissues in high concentrations and in normal tissues in low concentrations, this has not been the case so far. Instead of just targeting tumors, aptamers also accumulate in normal organs, particularly in the liver and kidneys. This normal-tissue accumulation is undesirable because it decreases the aptamers’ therapeutic effect in tumors and increases toxicity in these normal organs.
The researchers examined a specific aptamer type that binds with high affinity to nucleolin, a protein overexpressed in several cancers, including certain breast cancer cells. They engineered a complementary, light-sensitive oligonucleotide—a short, single DNA strand—that binds to the aptamer and deactivates it. In the presence of UV light, the complementary oligonucleotide’s bonds break down and release the now-active aptamer. This allows for greater control over distribution, because the aptamer is only activated where the UV light is shone; it remains inactive elsewhere.
Researchers validated their approach in cell lines and a murine model of breast cancer by introducing the hybridized aptamer/oligonucleotide complex and shining a UV light over the physical location of the tumor. In comparison to tests with the unbound, free aptamer, they found that the light-activated aptamer was distributed more favorably in the tumor as accumulation in the liver and kidney decreased. Furthermore, the light-activated aptamer targeted tumor tissues with similar accuracy and retention as the free aptamer.
This strategy holds promise for using light triggering to improve the accuracy and effectiveness of drug delivery, especially as aptamers can be attached to a variety of therapeutic agents and delivery devices, including chemotherapy drugs, siRNA, and nanoparticles. While UV light has limitations in its ability to penetrate tissue, the researchers suggest that leveraging infrared light—which has greater penetration depths—or introducing UV light via endoscopy are areas for further exploration.
“The importance of this research is in the demonstration of the spatiotemporal regulation of aptamer activity in vitro and in vivo that can be achieved by a photo-triggered strategy,” says Lele Li, one of the paper’s lead authors. “Our strategy may provide an approach to improving the therapeutic indices of aptamer-based medicines.”
The paper’s other lead author is Rong Tong. Both Li and Tong are postdocs in Langer’s and Kohane’s laboratories.