Image Credit: © Wiley-VCH, Angewandte Chemie.
But the fluorescent dyes utilized in such methods are always quite unstable, and poor pharmacokinetics, photobleaching, and lack of specificity are repeated problems.
Researchers from the United States have designed a molecular shield that improves the functionality of near-infrared fluorescent dyes by stabilizing them. The researchers have described the production and characterization of the molecular shield in the Angewandte Chemie journal.
In general, the near-infrared region of the spectrum is used for fluorescence bioimaging since this radiation can effectively penetrate through human tissue. Fluorescent dyes developed to serve this purpose often have a flat, symmetrical molecular architecture.
Although such a structure supports the absorption of near-infrared light, the dyes must be water-soluble and contain functional groups for combining with targeting biomolecules, for instance, tumor-binding peptides or antibodies.
Heptamethine cyanines, or Cy7, is a member of this group of fluorescent dyes currently under study in surgical applications.
But the Cy7 molecules have their disadvantages. They have a light-absorbing chromophore that is susceptible to oxygen radicals, resulting in bleaching. Moreover, the flat rigid molecules may accumulate and interact with other biomolecules nonspecifically, which decelerates their clearance from the body.
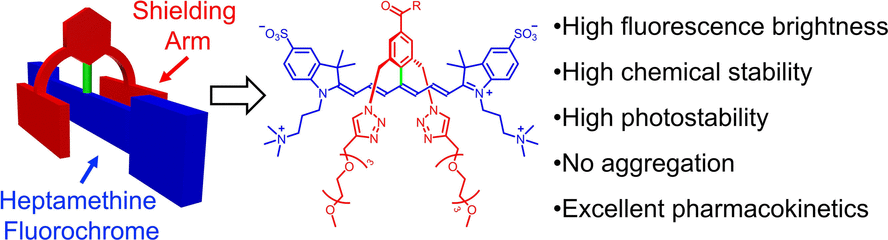
To overcome such problems, Bradley D. Smith together and his colleagues at the University of Notre Dame, USA, enhanced the dye’s chemical structure. To safeguard the heptamethine chromophore from oxygen attack, the research team added a smart and voluminous shield.
They fixed a heavy aromatic group on top of the chromophore’s central part and equipped this top group with long shielding arms that protruded over both faces of the chromophore, similar to how a bird covers its nest with its wings.
The ensuing dye was called a “sterically shielded heptamethine cyanine dye,” or s775z, by the researchers and was water-soluble and exhibited stable fluorescence. According to the authors, the shielded architecture avoided photobleaching and aggregation.
They also added that the dye was extremely firm against chemical degradation and could be stored “indefinitely” in a typical refrigerator.
Imaging studies by the researchers in live mice showed that in contrast to all other dyes analyzed, s775z did not aggregate in the blood clearance organs, but was eliminated from the body very fast. Furthermore, in tumors, a cancer-targeted version of s775z aggregated at a high level and could be imaged through fluorescence imaging of the live mice.
The researchers note that the new s775z dye can be utilized for an extensive range of biomedical imaging applications. They suggest the transformation from a flat molecule to a three-dimensional shielding architecture was crucial in making this family of near-infrared fluorescence dyes highly efficient and firm.
Journal Reference:
Li, D.-H., et al. (2020) Sterically Shielded Heptamethine Cyanine Dyes for Bioconjugation and High Performance Near-Infrared Fluorescence Imaging. Angewandte Chemie. doi.org/10.1002/anie.202004449.