Nov 1 2016
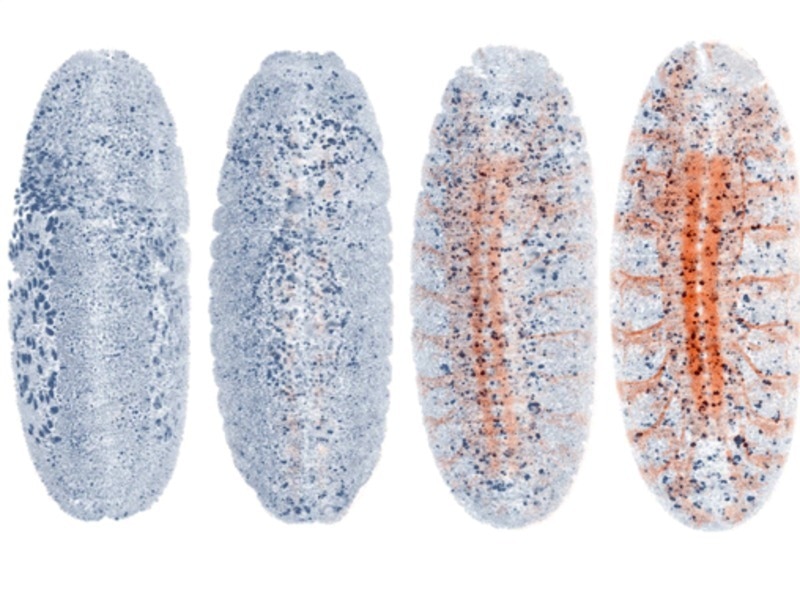 20-h imaging of a fruit fly embryo with the nervous system fluorescently labeled. AutoPilot automatically detects the onset of the expression of the marker and optimizes all parameters associated with this color channel in response to the emerging signal. © MPI of Molecular Cell Biology and Genetics.
20-h imaging of a fruit fly embryo with the nervous system fluorescently labeled. AutoPilot automatically detects the onset of the expression of the marker and optimizes all parameters associated with this color channel in response to the emerging signal. © MPI of Molecular Cell Biology and Genetics.
Effective imaging of the development and function of entire living organisms can be achieved by employing light-sheet microscopy. Yet, manual adjustments ought to be made during imaging to obtain high-resolution images using these microscopes. Scientists from the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden worked along with collaborators from the Janelia Research Campus (HHMI) to develop a new type of light-sheet microscope that can automatically “drive” on its own by adjusting itself to the dynamic and challenging optical conditions of large living organisms. This smart microscope is a combination of a smart ‘AutoPilot’ system, which is capable of automatically adjusting and improving the microscope to examine images, and a novel hardware model. For the maiden time, this construction allows long-term adaptive imaging of whole developing embryos and enhances the light-sheet microscope resolution up to five times.
Light-sheet microscopy, developed in the past ten years, is an innovative microscopy procedure specifically suitable for imaging large living organisms. A laser light sheet present in a light-sheet microscope illuminates the specimen perpendicular to the observation along a thin plane within the specimen. Since the observed plane is illuminated, the scattered and out-of-focus light from other planes (generally affecting image quality) is substantially prevented.
The long-time objective of microscopy is to attain in-depth ever-sharper images of living samples. To achieve this, the alignments between light-sheet illumination and imaging planes in the light-sheet microscopes ought to be carefully and perfectly maintained. However, misalignments of the planes occur due to the optical variability of living tissues over time and across disparate locations. Therefore, this challenge must be overcome to obtain high-resolution images that are mandatory to decrypt the biology related to organism development and morphogenesis.
So far, researchers had to sit at their microscope and tweak things manually—our system puts an end to this: it is like a self-driving car—it functions autonomously.
Loïc Royer, first author of the study
This autonomous smart microscope can examine and improve the spatial relationship between detection planes and light sheets across the sample volume in real time.
The scientists imaged the development of fly embryos and zebrafish for more than 20 hours to exhibit the performance of the smart autonomous microscope. They also carried out adaptive whole-brain functional imaging on larval zebrafish, thus acquiring highly sharp images of an entire “thinking” fish brain. In the research, they have displayed the way in which the framework recovers sub-cellular and cellular resolution in numerous regions of the specimen and also how it adjusts itself to the changes in the spatial distribution of fluorescent markers.
We have been using our AutoPilot system on several microscopes for more than two years and it makes a big difference in terms of image quality.
Philipp Keller, senior author
Constructing autonomous and adaptive microscopes is highly crucial in enabling the prospective use of light-sheet microscopy for developing mutant screens and automated high-throughput drug screens, and for the construction of developmental and anatomical atlases in different biological model systems.