 By Atif SuhailReviewed by Lexie CornerUpdated on Jun 19 2025
By Atif SuhailReviewed by Lexie CornerUpdated on Jun 19 2025As temperatures rise, you may hear more about ultraviolet (UV) light. But what exactly is it?
UV light is a part of the electromagnetic spectrum located between visible light and X-rays. It has a wavelength range of 10 to 400 nanometers and photon energies between 3 and 124 electron volts (eV).
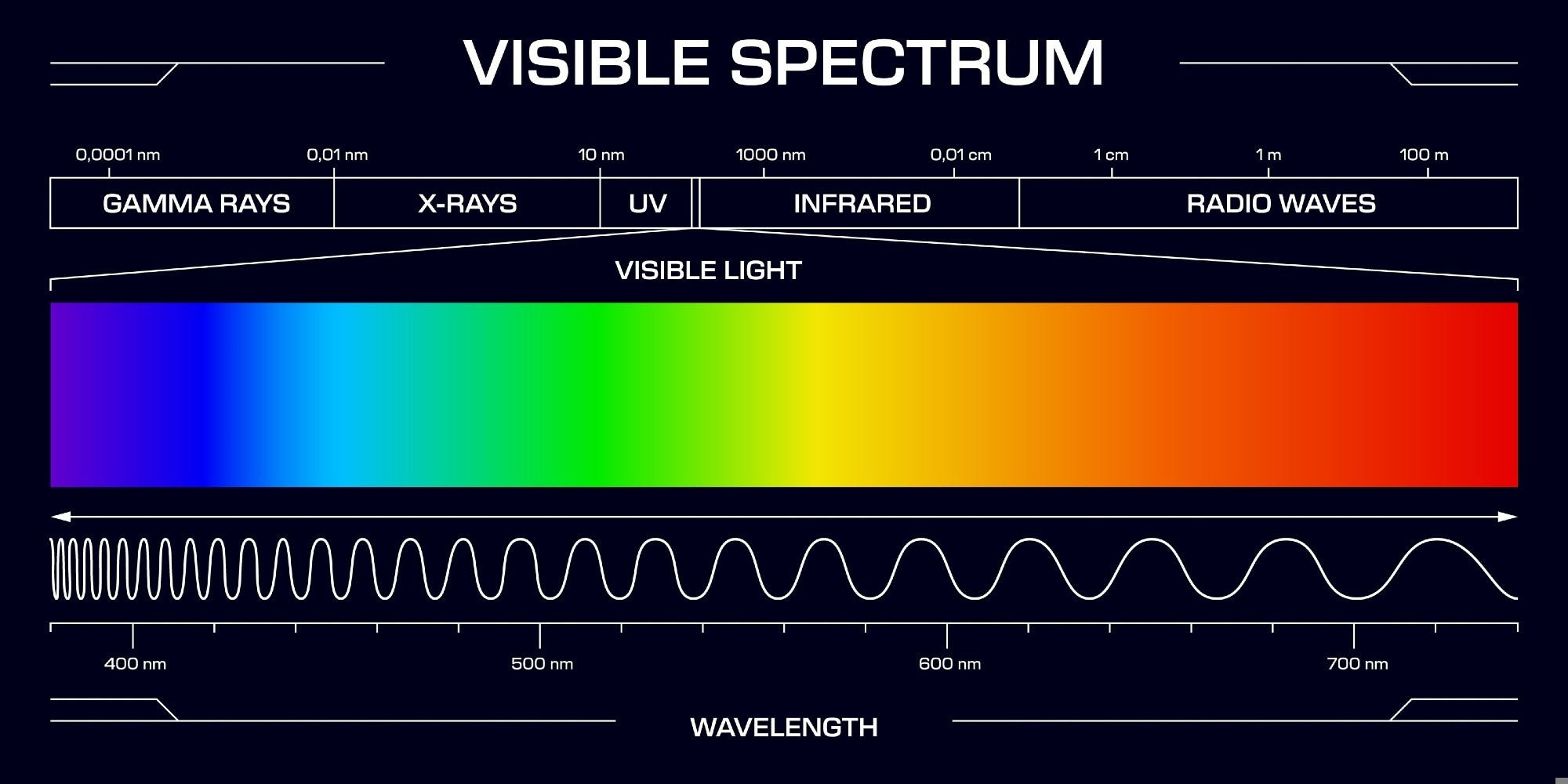 Image Credit: Buch and Bee/Shutterstock.com
Image Credit: Buch and Bee/Shutterstock.com
UV light was discovered in 1801 by German physicist Johann Wilhelm Ritter when he exposed silver salts to sunlight. Later, in 1893, another German physicist, Victor Schumann, identified vacuum ultraviolet (UV radiation with wavelengths below 200 nanometers).
What Are the Types of Ultraviolet Light?
UV light is invisible to the human eye and is classified into three main types based on wavelength: UV-A, UV-B, and UV-C. A fourth, emerging category is Far UV-C.
- UV-A (315–400 nm) has the longest wavelength. It is not absorbed by the Earth’s ozone layer and makes up the majority of UV radiation that reaches the surface. It penetrates deep into the skin.
- UV-B (280–315 nm) has a shorter wavelength and is mostly absorbed by the ozone layer—only about 5 % reaches the Earth's surface. It affects the outer skin layers, causing sunburn, but also helps the body produce vitamin D.
- UV-C (100–280 nm) has the shortest wavelength and is absorbed by the atmosphere, so it does not reach the ground under natural conditions.
- Far UV-C (200–230 nm), especially near 222 nm, is an artificial form of UV-C under study for safe disinfection in occupied spaces.
Where Does Ultraviolet Light Come From?
UV light originates from both natural and artificial sources.
The main natural source is the sun, which emits UV-A, UV-B, and UV-C radiation. However, the Earth’s ozone layer absorbs all UV-C and most UV-B, allowing only UV-A and a small portion of UV-B to reach the surface.
In space, stars—including our sun—produce UV radiation through high-energy reactions in their cores. This radiation contributes to processes such as gas ionization and star formation.
On Earth, artificial UV sources are designed to emit specific wavelengths for various applications. Common examples include:
- Black lights
- Short-wave UV lamps
- Gas-discharge and germicidal lamps
- Fluorescent and incandescent bulbs
- UV LEDs
- UV lasers
How Is UV Light Measured?
UV light is measured using a variety of detectors, depending on the specific range and application. Suitable sensors include photocathodes, photodiodes, and sensitive photomultipliers. For near and medium UV wavelengths, photographic films can also be used. Detection of vacuum UV typically requires wide-bandgap solid-state devices or vacuum-based instruments with high-cutoff photocathodes.
Three key parameters are typically involved in UV measurement:
- Wavelength: Measuring specific wavelengths of a UV source requires a spectrometer or spectral radiometer. These devices can identify individual peaks across the UV spectrum.
- UV irradiance: This refers to the power of UV radiation per unit area. It is essential for determining whether sufficient energy is delivered for processes like UV curing.
- UV energy density: Also known as dosage, this is the total energy received over time. It can be calculated by combining irradiance with the exposure duration.
In addition to scientific instruments and measurement parameters, the UV Index is a widely used tool that helps the public understand daily UV risk levels. Developed in 1987 with the World Health Organization, it translates complex UV data into an easy-to-read scale.
For a simple overview of how it works, this short video explains the origins of the UV Index and how sensors mimic the way our skin responds to sunlight:
UV-Index - What it is and how to measure it.
What Is Ultraviolet Light Used For?
UV light has a wide range of applications across healthcare, industry, science, and consumer products.
Medical Applications
UV-C light, particularly in the 200–280 nm range, is effective at inactivating bacteria, viruses, and other pathogens. It is commonly used in hospitals to disinfect surgical instruments, patient rooms, and air systems.
UV light also plays an important role in treating certain skin conditions. UVB phototherapy is widely used to manage psoriasis, eczema, and vitiligo by slowing the growth of affected skin cells.
In a clinical study, Gläser et al. found that UVB irradiation can stimulate the expression of antimicrobial peptides (AMPs) in human skin. Participants with Fitzpatrick skin types II and III were exposed to UVB light at a peak wavelength of 313 nm, with doses ranging from 250 to 1500 J/m2.
Industrial Applications
In manufacturing, UV curing is used to rapidly harden inks, adhesives, and coatings. This technique improves production efficiency and enhances the durability of finished products. Industries such as automotive, electronics, and printing commonly apply UV curing for protective coatings and adhesive bonding.
UV light is also used to treat surfaces, improving characteristics like scratch resistance and adhesion. For instance, UV-treated coatings on optical fibers increase their durability and performance.
In one study, Alcón et al. developed a UV-curable photoluminescent powder coating using unsaturated polyester resin, strontium aluminate pigment, and a specific photoinitiator. By adjusting extrusion and milling temperatures, they created an eco-friendly coating suitable for heat-sensitive substrates.
Scientific Applications
UV spectroscopy is an important analytical tool in chemistry and biology. It enables scientists to study how substances absorb and emit ultraviolet light, which aids in identifying chemical compounds and understanding molecular structures.
Click to Learn How to Interpret UV-Vis Spectroscopy Results
In astronomy, space-based telescopes equipped with UV detectors—such as the Hubble Space Telescope—observe celestial phenomena that are not visible in other parts of the spectrum. UV observations provide valuable insights into processes such as star formation, galaxy evolution, and the behavior of interstellar matter.
A recent study by Y. Shvartzvald et al. describes ULTRASAT, a space telescope scheduled for launch in 2027. It will perform the first wide-field near-ultraviolet (NUV) survey of transient and variable cosmic events.
With rapid data transmission and fast slewing capabilities, ULTRASAT is expected to support early detection of supernovae and quick follow-up of gravitational wave events, offering a new perspective on the dynamic, high-energy universe.
Consumer Applications
UV light is commonly used to verify the authenticity of banknotes, passports, and other official documents. Many of these items contain UV-reactive features that are only visible under ultraviolet illumination, helping to prevent counterfeiting.
In the beauty industry, UV nail lamps—typically emitting UV-A light—are widely used in salons to cure gel nail polish. This process quickly hardens the polish and provides a long-lasting finish. Although generally considered safe, it is recommended to take precautions such as applying sunscreen or wearing fingerless gloves to reduce skin exposure during use. UV-A is also used in black lights and tanning beds.
Is Ultraviolet Light Harmful?
UV light can be both beneficial and harmful, depending on the type of radiation, intensity, and duration of exposure. While UV light supports important health and technological functions, excessive or unprotected exposure carries serious health risks.
Prolonged exposure to UV-A and UV-B rays can cause immediate effects such as sunburn, as well as long-term damage, including premature skin aging, DNA mutations, and an increased risk of skin cancer. UV-B is particularly linked to melanoma and other forms of skin cancer, making it the most biologically active and potentially harmful part of solar UV radiation.
UV exposure also affects eye health. Over time, it can contribute to cataracts, macular degeneration, and photokeratitis—a painful condition similar to sunburn that affects the cornea. As with the skin, cumulative damage to the eyes increases with repeated exposure without proper protection.
Despite these risks, UV light—especially UV-B—also provides health benefits. The most well-known is the production of vitamin D. When UV-B rays interact with the skin, they initiate a chemical process that produces this essential nutrient, which supports bone health, immune function, and overall well-being. However, only brief and moderate exposure is needed for this benefit; excessive sun exposure is not necessary and is not recommended.
How to Stay Protected
Minimizing UV-related health risks is simple with the right precautions:
- Sunscreen: Use a broad-spectrum sunscreen with SPF 30 or higher on all exposed skin, even on cloudy days. Reapply regularly, especially after sweating or swimming.
- Protective Clothing: Wear wide-brimmed hats, long sleeves, and UV-blocking sunglasses to shield both skin and eyes from harmful rays.
- Limit Exposure: Stay out of direct sunlight during peak hours, typically from 10 a.m. to 4 p.m., when UV levels are at their highest.
- Use PPE in Work Environments: In settings where artificial UV light is used—such as medical sterilization or industrial curing—wear appropriate personal protective equipment (PPE), including face shields, gloves, and UV-blocking goggles.
References and Further Readings
- Guerrero, S. N.; Ferrario, M.; Schenk, M.; Fenoglio, D.; Andreone, A., Ultraviolet Light. Electromagnetic Technologies in Food Science 2021, 128-180. https://doi.org/10.1002/9781119759522.ch6
- Akash, M. S. H.; Rehman, K.; Akash, M. S. H.; Rehman, K., Ultraviolet-Visible (Uv-Vis) Spectroscopy. Essentials of pharmaceutical analysis 2020, 29-56. https://link.springer.com/chapter/10.1007/978-981-15-1547-7_3
- Correa, M. d. S. S.; Saavedra, M. e. R. R.; Parra, E. A. E.; Ontiveros, E. N.; Flores, J. d. C. B.; Montiel, J. G. O.; Contreras, J. E. C.; Urrutia, E. L.; Acevedo, J. G. Á.; Nopala, G. E. J., Ultraviolet Radiation and Its Effects on Plants. In Abiotic Stress in Plants-Adaptations to Climate Change, IntechOpen: 2023. https://www.intechopen.com/chapters/85608
- Ou, Y.; Petersen, P. M., Application of Ultraviolet Light Sources for in Vivo Disinfection. Japanese Journal of Applied Physics 2021, 60, 100501. https://iopscience.iop.org/article/10.35848/1347-4065/ac1f47
- Gläser, R.; Navid, F.; Schuller, W.; Jantschitsch, C.; Harder, J.; Schröder, J. M.; Schwarz, A.; Schwarz, T., Uv-B Radiation Induces the Expression of Antimicrobial Peptides in Human Keratinocytes in Vitro and in Vivo. Journal of Allergy and Clinical Immunology 2009, 123, 1117-1123. https://pubmed.ncbi.nlm.nih.gov/19342087/
- Czachor-Jadacka, D.; Pilch-Pitera, B., Progress in Development of Uv Curable Powder Coatings. Progress in Organic Coatings 2021, 158, 106355. https://www.sciencedirect.com/science/article/abs/pii/S0300944021002265
- Wali, A. S.; Kumar, S.; Khan, D., A Review on Recent Development and Application of Radiation Curing. Materials today: proceedings 2023, 82, 68-74. https://www.sciencedirect.com/science/article/abs/pii/S2214785322071930?via%3Dihub
- Alcón, N.; Tolosa, A.; Rodríguez, M.; Moreno, C., Development of Photoluminescent Powder Coatings by Uv Curing Process. Progress in Organic Coatings 2010, 68, 88-90. https://www.sciencedirect.com/science/article/abs/pii/S030094401000069X?via%3Dihub
- Krtička, J.; Benáček, J.; Budaj, J.; Korčáková, D.; Pál, A.; Piecka, M.; Zejda, M.; Bakış, V.; Brož, M.; Chang, H.-K., Science with a Small Two-Band Uv-Photometry Mission Ii: Observations of Stars and Stellar Systems. Space Science Reviews 2024, 220, 24.https://arxiv.org/abs/2306.15081
- Shvartzvald, Y.; Waxman, E.; Gal-Yam, A.; Ofek, E.; Ben-Ami, S.; Berge, D.; Kowalski, M.; Bühler, R.; Worm, S.; Rhoads, J., Ultrasat: A Wide-Field Time-Domain Uv Space Telescope. The Astrophysical Journal 2024, 964, 74. https://ui.adsabs.harvard.edu/abs/2024ApJ...964...74S/abstract
- Kurti, M.; He, Y.; von Lampe, K.; Li, Y., Identifying Counterfeit Cigarette Packs Using Ultraviolet Irradiation and Light Microscopy. Tobacco control 2017, 26, 29-33. https://pubmed.ncbi.nlm.nih.gov/26699918/
- Dowdy, J. C.; Sayre, R. M., Photobiological Safety Evaluation of Uv Nail Lamps. Photochemistry and Photobiology 2013, 89, 961-967. https://pubmed.ncbi.nlm.nih.gov/23550905/
Additional Sources
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.