Optical air monitoring technology offers remote, real-time pollution detection.

Image Credit: Anastasiia Tymoshenko/Shutterstock.com
Over the last century, the world’s population has grown rapidly. Consequently, industrial manufacturing plants have also expanded exponentially. A by-product of manufacturing is the emission of pollutants into the atmosphere.
Air Pollution
Examples of harmful gases found in the air are carbon monoxide (CO), carbon dioxide (CO2), methane (CH4), and nitrogen dioxide (NO2). Poisonous gases are typically created from combustion processes. The burning of wood, oil, coal, charcoal, natural gas, and other fossil fuels are some examples of combustion processes.
Exposure to high levels of air pollution can cause adverse health conditions, including respiratory infections, heart disease, and lung cancer.
CO2 emissions and other greenhouse gases have been correlated to global warming. CO2 absorbs infrared energy from the sun and other sources and re-emits the energy back in all directions. While a percentage of that energy radiates out into space, the rest of it is directed back onto earth. Thus, the more CO2 produced on earth, the more heat directed back onto earth.
Air Monitoring Methods
It is essential to monitor the quality of air and take mitigating actions to reduce air pollution. Many air quality monitoring methods have been developed over the years. Some examples include:
- Gas Chromatography (GC): A technique where components of the gas sample are dissolved in a solvent and vaporized to separate the constituents.
- Immunoassay: A sensing method that uses antibodies to identify and quantify organic compounds.
- Mass Spectrometry: An experimental analytical technique that differentiates organic compounds by measuring the mass of the compound's molecule.
- Ground Penetrating Radar (GPR): A geophysical technique used for investigations of the earth’s subsurface with high resolution, potentially recovering materials that can radiate gases into the environment.
While the techniques listed above are still widely used for specific sensing applications, monitoring methods based on optics have proven to be an ideal method for remote and real-time gas monitoring.
Molecular Absorption of Light
The basic concept of optical gas sensing is based on the molecular absorption of light, which can be described with infrared spectroscopy. Infrared (IR) spectroscopy is the interaction of IR light with matter. When IR light passes through a molecule, it can trigger vibrations on specific bonds within the molecule. This phenomenon of a molecule taking energy from the light is called absorption. The molecule only absorbs energy or photons at a specific wavelength of the IR beam and the energy absorbed by the molecule will no longer be detectable in the IR light beam.
Molecular absorption can be leveraged to set up a detection scheme for spectroscopy as shown in figure 1.
An artificial light source such as a laser or natural light rays from the sun can provide the source of incident light. The sample or the gas cloud is set in the path of the incident light. A small amount of light will be reflected or backscattered by the sample.
A larger fraction of the incident light will travel through the sample. While the light traverses the sample, some of the energy will be absorbed by the molecules in the sample. The part of the light that is transmitted and carries the sample’s molecular information can be collected by a detector to produce an electronic signal.
Similar information about the molecules can also be detected on the reflected beam. A typical spectrum produced by the detector will show a dip at the wavelength where the molecule absorbed energy from the light beam. For a particular molecule, for example, CO2, the dip will appear at a specific wavelength.
Through this process, a database of molecular “fingerprints” can be populated. This spectroscopy-based method of sensing gas molecules can be carried out on mobile platforms with data acquisition performed in real-time.
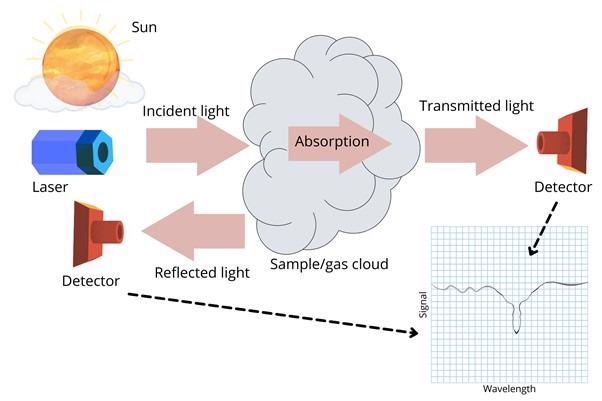
Figure 1: Illustration of an IR spectroscopy set-up. The Wavelength vs Signal plot on the bottom right shows a typical spectrum. Image Credit: Ilamaran Sivarajah
Optical Air Monitoring Techniques
There are other advanced optical techniques that have been developed for air pollution monitoring. Some of the optical methods used are:
- Ultraviolet-Differential Optical Absorption Spectroscopy (UV-DOAS): A technique similar to IR spectroscopy that uses ultraviolet, visible, and near-infrared spectrum to identify and quantify individual chemicals.
- Laser-Induced Fluorescence (LIF): A laser-based technique that can provide highly detailed information about the distribution of contaminants.
- Fiber Optic Chemical Sensors (FOCS): The light is delivered to the detection location by fibers. The wavelength or intensity changes of the laser provide information about the environment.
- Fourier Transform Infrared (OP-FTIR) spectroscopy: A versatile technique capable of measuring the presence of many chemicals in the air simultaneously with high sensitivity.
- LIDAR: This is based on radar, an established technology that uses radio waves. Lidar operates on the same principles as radar except that it uses light to collect information.
How is Air Quality Data Used?
The various methods described above form an integral part of an effective air quality management system. Real-time, remote sensing can rapidly assess the extent of pollution to prevent catastrophic consequences in circumstances such as gas leaks.
In general, air quality monitoring helps gather air pollution data to set air quality goals or standards. The data collected is also used to evaluate the effectiveness of emissions control strategies, provide information on air quality trends, and support research such as long-term studies of the health effects of air pollution.
References and Further Reading
Feng S, Farha F, Li Q, Wan Y, Xu Y, Zhang T, Ning H. Review on Smart Gas Sensing Technology. Sensors. 2019; 19(17):3760. https://doi.org/10.3390/s19173760
European Commission. Measuring air pollution with low-cost sensors | europa.eu https://publications.jrc.ec.europa.eu/repository/handle/JRC107461
Jihyun Kim, Joohyung Kim, and Seo Hyun Jang "Infrared spectra in air pollution research and monitoring from space: a review", Proc. SPIE 11590, Nano-, Bio-, Info-Tech Sensors and Wearable Systems, 115900P (8 April 2021); https://doi.org/10.1117/12.2584500
The European Space Agency. Sentinel-5 | sentinel.esa.int https://sentinel.esa.int/web/sentinel/missions/sentinel-5
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.