Skin, the largest organ of the body, possesses an incredible ability to heal itself even after significant damage.
As our principal barrier against microbes and the elements, it is crucial that we understand the dermal repair process and how to accelerate it when necessary. OCT provides a unique and non-destructive technique to do so, looking closely beneath the skin to depths of 1-2 mm, and with better than 10 µm resolution.
To test the potential of this, a group headed by Irena Pastar at the University of Miami resolved to assess the use of OCT as an alternative method to tissue sectioning in wound healing studies. The result? OCT demonstrated the ability to capture detailed wound images competing with those acquired via microscopy and produced a better objective assessment of the healing advantages gained from a new stem cell therapy.
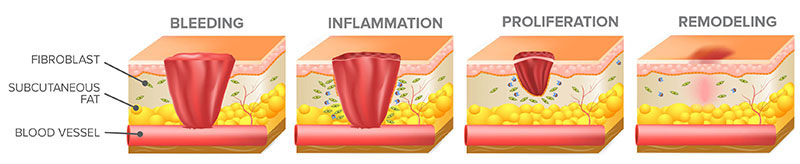
The wound healing process involves overlapping stages of bleeding, inflammation, proliferation, and remodeling (epithelialization).
Revealing the Healing Process
Throughout the wound healing process, the skin experiences interlacing phases of inflammation, proliferation (or migration), and remodeling. While the action of acute wound healing hasn’t been examined in-vivo where humans are concerned, for ethical reasons, it is possible to closely mimic this process, ex-vivo.
Using donated skin from reduction surgeries, the ‘ex-vivo’ model for wound healing is conducted under conditions supervised and controlled long enough to examine the molecular mechanisms involved and to assess potential therapies intended to accelerate healing.
The main disadvantage of this model is that assessment has typically required the destruction of the wounds to assess the healing process via histologic examination (HE). Several wounds that are identical must be made to study a single wound type or therapy. Subsequently, careful sectioning and staining of each wound is conducted in turn for examination under a microscope at different time points to see to what extent the epithelial tongue has grown from the wound edge to cover the exposed dermis.
Comparatively, OCT, permits the same wound to be observed non-destructively from beginning to end, eradicating variability between tissue samples and individual wounds. This is especially relevant given that wounds typically heal unevenly, closing rapidly at some edges more than others.
OCT can be used for the rapid collection of longitudinal images in cross-section that display dermal sub-structure, from which 3D volumes or en-face images of the whole wound can be produced. This promotes a more objective view of the wound as it heals, rather than being restricted to a particular cross-section and the quality of sample preparation.
Evaluating OCT for ex-vivo
Throughout this study, both swept-source (SS-OCT) imaging at ~1300 nm and spectral-domain (SD-OCT) imaging at ~850 nm were assessed for use in the evaluation of cutaneous wound healing. While both systems held the capacity to image to similar depth (~2 mm and ~1.9 mm, respectively), the SD-OCT system from Wasatch Photonics provided superior axial resolution (<3 µm) and transverse resolution (<6 µm) in contrast to the clinical SS-OCT system. Shorter operating wavelengths yield higher resolution OCT images, enabling improved differentiation of the different tissue types within the dermal structure.
In order to compare OCT imaging to histologic examination (HE), a series of identical wound samples were prepared, a portion of which was treated with rhEGF (recombinant epidermal growth factor). All samples were incubated at 37 °C, and triplicate samples were acquired for evaluation at days 0, 4, and 7 utilizing each method.
Wounds to be assessed by HE were set in formalin solution, placed in paraffin, and then sectioned in 8 µm slices for staining and microscopy; sampling from the wound’s edge of to its widest point in diameter. Progress of healing quality was measured by taking the sum of the epithelial tongues migrating from the wound edge and comparing it to the total wound diameter as a percentage.
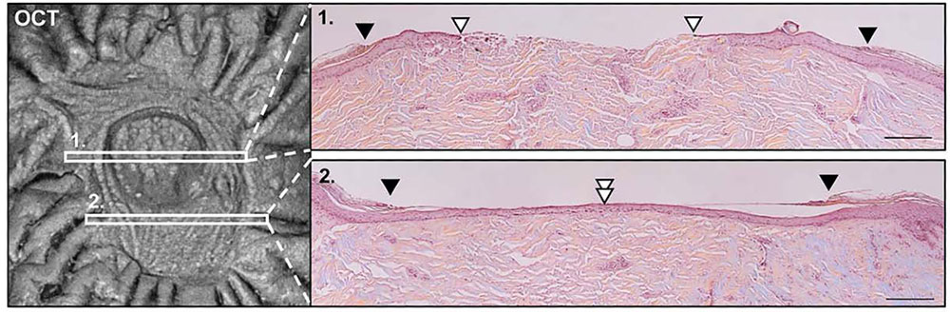
Comparison of 1300 nm SS-OCT en-face imaging to cross-sectional histologic analysis at two points on a healing wound.
For the OCT analysis, samples could be assessed and measured in cross-section from one wound edge to the other in less than 1 minute, building up en-face wound images of the tissue surface from the longitudinal scans. In these ‘top-down’ views of the wound, the unbroken epidermis could be easily discerned from the migrating epithelial tongue as it reached to cover the exposed dermis at the center. This permitted a better objective area calculation to be utilized in order to quantify healing in terms of percent of wound healed vs the original wound area.
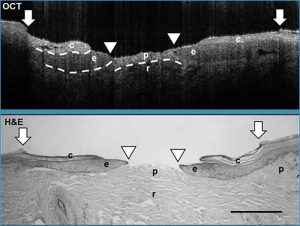
Visualization of tissues in ex vivo human wounds with (a) histologic evaluation and (b) using a Wasatch Photonics 800 nm OCT system (c). Depicted are c= cornified epidermis; e= noncornified epithelial tongue; p= papillary dermis; r= reticular dermis.
OCT regularly demonstrated a higher degree of re-epithelialization (healing) in contrast to HE analysis, which is no wonder given its capacity to image the full wound vs extrapolating from cross-sections. Nevertheless, the data illustrated a powerful positive correspondence between OCT and HE analysis, and an almost flawless agreement for both new and fully healed wounds, thus validating OCT as a workable methodology for analysis.
Cross-sections acquired using the 800 nm SD-OCT system from Wasatch Photonics offered adequate resolution and contrast to easily determine the epidermis, the papillary, and reticular dermis, and the migrating epithelium in the wound bed. The applicability of OCT in dermatology extends well beyond an evaluation of wound healing or novel therapies, due to the capacity to differentiate between tissue types it extends to studies of the mechanisms of stem cell action and epidermal migration during re-epithelialization. OCT’s ability to provide rapid, reproducible scans non-invasively holds great promise for use in preclinical testing, and for guiding in-depth immunohistochemistry analyses by determining the location for optimal sectioning.
Wound Healing as Observed by OCT
Longitudinal cross-sections acquired using 850 nm SD-OCT at 0, 4, and 7 days postwounding, imaging from one wound edge to the other in ~10 µm increments, collected at 22 frames per second.
A Novel Stem Cell Therapy
Pastar’s group implemented the latest imaging modality to analyze the therapeutic potential of allogeneic human adipose-derived stem cells (ASC’s) in human ex vivo wounds after confirming OCT was a viable ‘optical biopsy’ alternative to histologic analysis.
Stem cell therapies have demonstrated promise for the treatment of difficult wounds, but their use can be a contentious issue. Stem cells taken from adult adipose (fatty) tissue, tend to be free of this ethical constraint. Furthermore, they have been shown to facilitate various aspects of wound healing, from differentiation to vasculogenesis.
Along with the previous validation study detailed earlier, a series of ex vivo wounds were treated with ASC’s via an injection into the middle of the fresh wound. These wounds were also checked with OCT over the exponential phase of healing in ex-vivo wounds – 4 days.
Using OCT imaging, it was discovered that all ASC-treated wounds had re-epithelialized completely, while media treated control wounds showed only 50% wound closure. The difference in healing for the ASC-treated wounds vs rhEGF-treated wounds was observed to be statistically significant, and the increase of wound closure was affirmed via cross-sectional OCT images that determined both single layer and differentiated layers of epidermis.
The capacity of allogeneic human ASC to encourage re-epithelialization for 3 mm diameter wounds extending to the dermis, necessitated a single ASC treatment which resulted in significant stimulation of healing, as recorded by OCT.
Given the case history of the ex-vivo wound model for assessing unique therapeutics, and the capacity of OCT to be employed in preclinical studies, the assessment of ASC therapy with OCT for in-vivo use in wound treatment fosters great potential.
Conclusion
OCT has demonstrated both its superiority and practicability for non-invasive observations of human wound healing: the conclusion reached by imaging ex-vivo wounds non-destructively to 2 mm depths and better than 10 µm resolution. Moreover, not only is it more consistent and faster than histologic examination, but it also enables continuous monitoring throughout the wound healing process while providing appropriate resolution to evaluate both anatomy and pathology.
This makes it a method that puts forward promising solutions for applications in wound healing and the assessment of novel therapeutics in both laboratory and clinical settings. When employed to study treatment of wounds with ASC’s, OCT showed a considerable increase in re-epithelialization after just four days, further demonstrating OCT’s potential to make a valuable contribution to the field of wound healing.
References and Further Reading
Glinos, George D., et al. “Optical coherence tomography for assessment of epithelialization in a human ex vivo wound model.” Wound Repair and Regeneration 25.6 (2017): 1017-1026.

This information has been sourced, reviewed and adapted from materials provided by Wasatch Photonics, Inc.
For more information on this source, please visit Wasatch Photonics, Inc.